Arabidopsis Research Roundup: March 17th
This weeks UK Arabidopsis Research Roundup includes three papers featuring researchers from the University of Nottingham as well as manuscripts from Leeds, Lancaster, QMUL and The Sainsburys Lab in Norwich
Firstly Stefan Kepinski (Leeds) leads a study that investigates how Gravitropic Set Point Angle (GSA) is controlled in response to different growth factors. Secondly are two Methods papers featuring researchers from CPIB in Nottingham, the first of which is in collaboration with Lancaster University and introduces the Microphentron, which is an automated phenotyping platform that can be used for chemical biology screens. The second paper describes a non-destructive method for imaging floral tissues using CT scanning.
Ranjan Swarup is also a member of CPIB and in the next paper he has collaborated with French colleagues to investigate the role of SHR on root development in rice.
The fourth paper includes Cyril Zipfel as a co-author and investigates the role of damage-associated molecular patterns (DAMPs) in the response to pathogen attack whereas this weeks final paper is from the lab of Alexander Ruban (QMUL) and discovers the phenotypic consequences of persistent damage to PSII by photoinhibition.
Suruchi Roychoudhry, Martin Kieffer, Marta Del Bianco, Che-Yang Liao, Dolf Weijers Stefan Kepinski (2017) The developmental and environmental regulation of gravitropic setpoint angle in Arabidopsis and bean Scientific Reports http://dx.doi.org/10.1038/srep42664
Open Access

Stefan Kepinski (University of Leeds) leads this study that involves a collaboration with Dolf Weijers from Wageningen University. They investigate the role of both auxin and environmental factors in determining gravitropic set point angle (GSA), which is a measure of the growth of lateral organs away from primary shoots and roots. They show that nitrogen and phosphorous deficiency causes opposing effects on lateral root GSA, each of which are auxin-dependent. This contrasts with previous findings from work using bean adventitious roots. They find that these differences are maintained when Arabidopsis and bean roots are treated with different auxin concentrations. Latterly they also look at the effect of different light conditions on shoot GSA and put these findings into the context of potentially altering crop growth.
Stefan takes some time to discuss this paper for the GARNet YouTube Channel.
Burrell T, Fozard S, Holroyd GH, French AP, Pound MP, Bigley CJ, James Taylor C, Forde BG (2017) The Microphenotron: a robotic miniaturized plant phenotyping platform with diverse applications in chemical biology. Plant Methods
http://dx.doi.org/10.1186/s13007-017-0158-6 Open Access
This methods paper is a collaboration between the Universities of Lancaster and Nottingham led by Brian Forde that describes the Microphenotron. This device has been developed to facilitate chemical biology screens on in vivo plant tissues. This allows for the automated screening of either dicot or monocot roots or aerial tissues that have been grown on media infused with whichever chemical is relevant for the intended expriments. In situ GUS screening is also possible allowing for researchers to integrate information about growth and gene expression. The use of ‘Phytostrips’ in a 96-well format allows for high-throughput screening that is aligned with AutoRoot automated image analysis software to provide a rapid and facile method for undertaking small scale phenotypic screens. The Microphenotron facility is housed at the Lancester University, who are extremely open to collaboration so please get in contact if you are interested in using the facility.

Tracy SR, Gómez JF, Sturrock CJ, Wilson ZA, Ferguson AC (2017) Non-destructive determination of floral staging in cereals using X-ray micro computed tomography (µCT) Plant Methods. http://dx.doi.org/10.1186/s13007-017-0162-x Open Access
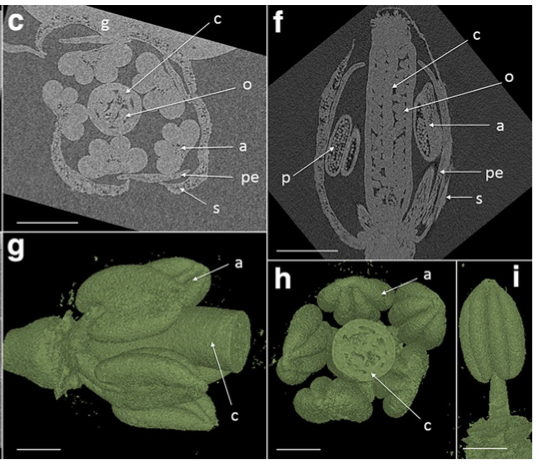
Alison Ferguson is the corresponding author on this methods paper that includes GARNet committee member Zoe Wilson and Saoirse Tracy from Dublin. They have developed a technique using X-ray µCT scanning to image developing flowers in Arabidopsis and barley plants, taking advantage of the excellent Hounsfield facility at the University of Nottingham. They show that the technique can be hugely beneficial for plant phenotyping by providing a non-destructive method of analyzing live floral development and how this can response to changes in the growth environment. Members of the Hounsfield facility are happy to discuss any potential collaborative work and future access to these type of facilities will hopefully be improved through the UKs involvement in the pan-european EMPHASIS project.
Henry S, Dievart A, Fanchon D, Pauluzzi G, Meynard D, Swarup R, Wu S, Lee CM, Gallagher K, Périn C (2017) SHR overexpression induces the formation of supernumerary cell layers with cortex cell identity in rice. Dev Biol. http://dx.doi.org/10.1016/j.ydbio.2017.03.001
Ranjan Swarup (CPIB) is a co-author on this study that includes French and US researchers. Previously they had shown that expression of rice SHORTROOT (OsSHR) genes could compliment the Arabidopsis shr mutant. In this study they show that overexpression of OsSHR and AtSHR in rice roots causes growth of wider, shorter roots that have an increased number of cortical cell layers. This demonstrates that the mechanisms that control the differentiation of cortical cell layers is conserved throughout land plants, with SHR being a key determinant in this process.
de Azevedo Souza C, Li S, Lin AZ, Boutrot F, Grossmann G, Zipfel C, Somerville S (2017) Cellulose-derived oligomers act as damage-associated molecular patterns and trigger defense-like responses. Plant Physiol. http://dx.doi.org/10.1104/pp.16.01680
Cyril Zipfel (The Sainsbury Lab) is a co-author on this study from the lab of Shauna Somerville in California that focuses on the concept of damage-associated molecular patterns (DAMPs). These can be defined as cell wall breakdown components and stimulate the same defence responses as more fully characterised pathogen- or microbe-associated molecular patterns (PAMPs). Intuitively this makes sense as during infection many pathogens will cause cell wall breakdown. The authors show that cellulose-derived oligomers trigger a signalling response similar to that caused by oligogalacturonides or chito-oligomers but that lacks an increase in ROS or in callose deposition. These results confirm that cellulose-derived signals feed into the plants mechanism for cell wall scanning and acts synergistically with other signals that result from pathogen attack.
Tian Y, Ungerer P, Zhang H, Ruban AV (2017) Direct impact of the sustained decline in the photosystem II efficiency upon plant productivity at different developmental stages. J Plant Physiol. http://dx.doi.org/110.1016/j.jplph.2016.10.017
Alexander Ruban (QMUL) leads this Sino-UK collaboration that investigates how the photoinhibiton of photosystem II impacts overall plant growth. In this study they use lincomycin to block chloroplast protein synthesis, which prevents the plant from restoring PSII function after photoinhibitory damage. Treated plants accumulate less starch and showed reduced above-ground biomass. This leads to a decrease in seed yield. Perhaps unsurprisingly this research shows that restoring the full function of PSII after photoinhibition to key to maintaining normally functioning electron transport rate that leads into metabolic production and growth rate.
Podcast: Play in new window | Download

Leave a Reply